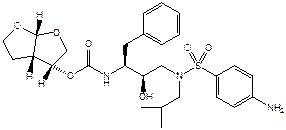Lord Justice Floyd:
1.
This appeal concerns the meaning of “the product is protected by a basic
patent in force” in Article 3(a) of Regulation (EC) No 469/2009 concerning the
supplementary protection certificate for medicinal products (“the SPC
Regulation”). The SPC Regulation regulates the grant of supplementary
protection certificates or SPCs which are instruments which, in effect, extend
the term of granted patents beyond their originally allotted term in relation
to a particular product which is the subject of a marketing authorisation.
Their purpose is to compensate the patent proprietor for the time which it
takes between filing a patent application and bringing a medicinal product to
the market. This issue of interpretation of the SPC Regulation has been the
subject of repeated references to the CJEU from the courts of the United
Kingdom and elsewhere, without, thus far, the emergence of a clear legal
criterion for national courts to apply in all cases.
2.
The first respondent is the proprietor (and the second respondent the
exclusive licensee) of SPC/GB07/038 for a product described in the SPC as
“Darunavir or the pharmaceutically acceptable salt, ester, or prodrug
thereof”. The SPC covers a product marketed in Europe by companies associated
with the second respondent under the trade mark “Prezista”. It is a protease
inhibitor used in an anti-retroviral medication for the treatment of the HIV
virus and AIDS. The respondents contend that the product described in the SPC
was protected by European Patent (UK) No 0 810 209 (“the patent”) of which the
first and second respondents were again, respectively, the proprietor and the
exclusive licensee.
3.
The SPC will expire on 23 February 2019. The appellants brought the
present proceedings in order to clear the way for the marketing of a generic
darunavir product prior to the expiry of the SPC. The appellants’ product is
not yet on the market. It is common ground, at least for the purposes of these
proceedings, that the marketing of the appellants’ product would infringe the
SPC, if the SPC is valid. The appellants contend that it is invalid because,
on the true construction of Article 3(a) of the SPC Regulation, darunavir is
not a product “protected” by the patent. There is no challenge to the validity
of the patent itself.
4.
In a decision dated 3 May 2017, Arnold J decided that darunavir was a
product protected by the patent. He declined to refer questions to the CJEU on
the interpretation of Article 3(a) of the SPC Regulation because he considered
that, on all tenable constructions of Article 3(a), darunavir was protected by
the patent. This is an appeal from that decision and his consequent order.
5.
On the appeal the appellants were represented by Ms Charlotte May QC and
Mr William Duncan. The respondents were represented by Mr Thomas Mitcheson
QC and Mr Stuart Baran.
The SPC
Regulation and the Explanatory Memorandum
6.
The SPC Regulation was preceded by the Commission’s Explanatory
Memorandum COM 90 101 on the proposed Regulation (“the Memorandum”), a document
which it is common ground is admissible as an interpretive aid to the
Regulation. The Memorandum made the point that the Regulation was intended to
be “simple and transparent”, not to “lead to excessive bureaucracy” and to be
capable of implementation without “an excessive administrative burden being
placed on” patent offices or the parties, or there being any requirement for
the creation of a new administrative body. In particular, it was foreseen that
“examination of the conditions to be fulfilled for the certificate to be granted
involves the use of objective data that are easy to verify.”
7.
The Memorandum also made it clear that it was all types of research
which were to be protected, not merely that part of the research programme
which moved from the discovery of a structure-activity relationship to the
individual compound: see e.g. paragraph 29:
“The proposal does not provide for any exclusions. In other
words, all pharmaceutical research, provided that it leads to a new invention
that can be patented, whether it concerns a new product, a new process for
obtaining a new or known product, a new application of a new or known product
or a new combination of substances containing a new or known product, must be
encouraged, without any discrimination, and must be able to be given a
supplementary certificate of protection provided that all of the conditions
governing the application of the proposal for a Regulation are fulfilled.”
8.
The Memorandum suggests, at paragraph 39, and albeit in the context of
Article 4, that a patent which protects a series of compounds based on a
formula can be used as the basis for the SPC:
“It is thus often the case in the chemical and pharmaceutical
field that a patent protects a series of products based on the same formula.
However, only some of these products will subsequently be developed and
possibly only one may be put on the market. In such a case, the certificate
will only protect the product covered by the authorization and not all of the
products protected by the patent.”
9.
The recitals of the SPC Regulation which are material are set out below:
[3] Medicinal products, especially those that are the result
of long, costly research will not continue to be developed in the Community and
in Europe unless they are covered by favourable rules that provide for
sufficient protection to encourage such research.
[4] At the moment, the period that elapses between the filing
of an application for a patent for a new medicinal product and authorisation to
place the medicinal product on the market makes the period of effective
protection under the patent insufficient to cover the investment put into the
research.
[5] This situation leads to a lack of protection which
penalises pharmaceutical research.
[6] There exists a risk of research centres situated in the
Member States relocating to countries that offer greater protection.
[7] A uniform solution at Community level should be provided
for, thereby preventing the heterogeneous development of national laws leading
to further disparities which would be likely to create obstacles to the free
movement of medicinal products within the Community and thus directly affect
the functioning of the internal market.
[8] Therefore, the provision of a supplementary protection
certificate granted, under the same conditions, by each of the Member States at
the request of the holder of a national or European patent relating to a
medicinal product for which marketing authorisation has been granted is
necessary. A regulation is therefore the most appropriate legal instrument.
…
[10] All the interests at stake, including those of public
health, in a sector as complex and sensitive as the pharmaceutical sector
should nevertheless be taken into account. …
10.
Articles 1, 3, 4 and 5 of the SPC Regulation provide, so far as
relevant:
Article 1
Definitions
For the purpose of this Regulation:
(a) ‘medicinal product’ means any substance or combination of
substances presented for treating or preventing disease in human beings or
animals and any substance or combination of substances which may be
administered to human beings or animals with a view to making a medical
diagnosis or to restoring, correcting or modifying physiological functions in
humans or in animals;
(b) ‘product’ means the active ingredient or
combination of active ingredients of a medicinal product;
(c) ‘basic patent’ means a patent which protects a product as
such, a process to obtain a product or an application of a product, and which
is designated by its holder for the purpose of the procedure for grant of a
certificate;
…
Article 3
Conditions for
obtaining a certificate
A certificate shall be granted if, in the Member State in
which the application referred to in Article 7 is submitted and at the date of
that application:
(a) the product is protected by a basic patent in force;
(b) a valid authorisation to place the product on the market
as a medicinal product has been granted in accordance with Directive 2001/83/EC
or Directive 2001/82/EC, as appropriate;
(c) the product has not already been the subject of a
certificate;
(d) the authorisation referred to in point (b) is the first
authorisation to place the product on the market as a medicinal product.
…
Article 4
Subject matter of
protection
Within the limits of the protection conferred by the basic
patent, the protection conferred by a certificate shall extend only to the
product covered by the authorisation to place the corresponding medicinal
product on the market and for any use of the product as a medicinal product
that has been authorised before the expiry of the certificate.
Article 5
Effects of the
certificate
Subject to the provisions of Article 4, the certificate shall
confer the same rights as conferred by the basic patent and shall be subject to
the same limitations and the same obligations.”
11.
It is common ground in the present case that the SPC satisfies the
conditions specified in Article 3 (b), (c) and (d).
The patent
12.
The patent is entitled "Alpha- and beta-amino acid
hydroxyethylamino sulfonamides useful as retroviral protease inhibitors". It
claims a priority date of 25 August 1992. The specification begins by stating
at [0002] that the invention relates to such inhibitors, and in particular to
"sulfonamide-containing hydroxyethylamine protease inhibitor compounds, a
composition and the use thereof for preparing a medicament for inhibiting
retroviral proteases such as human immunodeficiency virus (HIV) protease and
for treating a retroviral infection e.g. an HIV infection".
13.
The specification explains at [0003] that replication of a virus such as
HIV involves a stage in which certain gene products are translated into
proteins which are then processed by a retroviral protease. Inhibition of the
retroviral protease may inhibit viral replication. After acknowledging certain
prior art, the specification sets out a description of the invention at [0008]:
"The present invention is
directed to virus inhibiting compounds and compositions. More particularly, the
present invention is directed to retroviral protease inhibiting compounds and
compositions, to the use of such compounds for preparing medicaments for
inhibiting proteases, especially for inhibiting HIV protease and for treating a
retroviral infection such as HIV infection and for treating AIDS, to processes
for preparing the compounds and to intermediates useful in such processes. The
subject compounds are characterized as sulfonamide-containing hydroxyethylamine
inhibitor compounds."
14.
The detailed description of the invention, starting at [0009], includes
a series of paragraphs corresponding to the claims. These are framed by
reference to two formulae, Formula I and Formula II. Formula I is shown below.

15.
Formula I identifies the essential backbone of the claimed class of
compounds, and a number of variable substituents, shown as P1, P2,
R2, R3 and R4. In addition to identifying the
elements of structure, the formula shows a particular stereochemical
orientation. The specification lists broad ranges of possible values of P1,
P2, R2, R3 and R4. Preferred
compounds of Formula I are described at [0010]-[0012], with increasing levels
of specificity. 41 specific compounds are listed in [0012].
16.
Formula II is the same as Formula I except that it omits some of the
stereochemistry. The specification again lists ranges of substituent groups
and preferred compounds at [0013]-[0017].
17.
Prof Adlington, the appellants’ expert organic chemist, estimated that
the number of compounds covered by claim 1 of the patent was somewhere between
7 x 10135 and 1 x 10377. There were something of the
order of 8 x 1036 possibilities for the substituent P1
alone. By contrast the number of compounds specifically disclosed was, in his
opinion, approximately 100. No attempt was made to challenge these numbers in
the court below.
18.
It is explained at paragraph [0020] that compounds of the invention can
be used to prepare pharmaceutical compositions useful for treating retroviral
infections, in particular HIV or AIDS. The patent states at [0091] that the
compounds of the invention are effective antiviral compounds and effective HIV
protease inhibitors.
19.
It is common ground that there is no reference to darunavir anywhere in
the specification.
20.
Claim 1 of the patent covers a class of compounds defined by means of
Formula I. Claim 5 is an independent claim based on Formula II but with a
narrower list of substituents. Claims 2, 10 and 11 contain narrower lists of
substituents.
21.
The form of claim adopted in the present case is a familiar one in
chemical patents. It is based on a structural formula having a fixed element
with variable substituents to be chosen from amongst a defined class. Such a
formula is known as a Markush formula, a term which, as the judge explained, is
a term which originated in a 1924 decision of the Commissioner of Patents of
the United States Patent and Trade Mark Office, ex parte Markush 1925
C.D. 126, 340 O.G. 839 (Comm’r Pat 1924). The Markush formula enables a large
class of compounds to be claimed without the necessity of writing out every
single chemical entity. The use of a Markush formula in a claim is an
appropriate means of claiming an invention where the patentee’s invention has
involved the discovery of a new technical effect which he predicts will be
common to all members of the claimed class provided they share a common
structural element (e.g. in the present case, the backbone structural element
which is not permitted to vary in accordance with Formula I or II). Claims
relying on a Markush formula to define their scope are referred to as Markush
claims. They avoid the necessity of writing out in extenso every
possible member of the claimed class. A danger with such claims is that they
may cover compounds which do not show the claimed activity, and so result in
insufficiency under Article 83 of the European Patent Convention (EPC), or equivalent
national laws.
22.
The judge found that the practice of permitting the use of a Markush
formula in a patent claim had been followed by patent offices worldwide, and in
particular by the United Kingdom and the EPO. The UK Intellectual Property
Office’s Manual of Patent Practice, says that Markush claims:
“… are often used in chemical cases as a way of setting out
various functionally equivalent alternatives in one or more parts of the
chemical compound being claimed”
Darunavir
23.
The structural formula of darunavir is shown below:
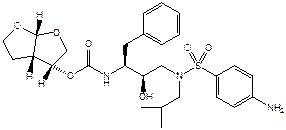
24.
The judge set out a helpful table to show how darunavir was a compound
represented by Formula I and Formula II within claims 1, 2, 5, 10 and 11 in
which the variable substituents were given values as follows:
|
Group
|
Claim 1
|
Claim 2
|
Claim 5
|
Claim 10
|
Claim 11
|
Darunavir
|
|
P1
|
Heterocyclyl-oxycarbonyl
|
Heterocyclyl-oxycarbonyl
|
Heterocyclyl-oxycarbonyl
|
Heterocyclyl-oxycarbonyl
|
Heterocyclyl-oxycarbonyl
|
Bis-THF derivative
|
|
P2
|
Hydrogen
|
Hydrogen
|
Hydrogen
|
Hydrogen
|
Hydrogen
|
Hydrogen
|
|
R2
|
Aralkyl
|
Benzyl
|
Aralkyl
|
Aralkyl
|
Benzyl
|
Benzyl
|
|
R3
|
Alkyl
|
Isobutyl
|
Alkyl
|
Alkyl
|
Isobutyl
|
Isobutyl
|
|
R4
|
Aryl
|
Para-substituted aryl
|
Aryl
|
Aryl
|
Para-substituted aryl
|
Para-amino-phenyl
|
25.
The appellants’ evidence was that the closest compound to darunavir
disclosed in the patent was the seventh compound listed in paragraph [0012]
(“Compound 7”). The difference between Compound 7 and darunavir is only in the
P1 substituent, which, in the case of Compound 7 is an
aralkoxycarbonyl group, specifically benzyloxycarbonyl. In darunavir the
corresponding P1 substituent is a heterocyclyloxycarbonyl group,
specifically a fused bis-tetrahydrofuran derivative. The difference in the two
P1 substituents is illustrated below, with that for Compound 7 on
the left and that for darunavir on the right:


26.
The appellants’ evidence was that the heterocyclyloxycarbonyl
substituent in darunavir was unusual, and not one which their expert had
previously encountered. The appellants’ case is that such a substituent does
not form part of the common general knowledge available to the skilled team at
the priority date. The appellants’ evidence was that the structure of the P1
substituent group of darunavir was not published until after the priority date
of the patent, in a paper by Arun K. Ghosh et al entitled “Structure-Based
Design of HIV-1 Protease Inhibitors: Replacement of Two Amides and a
10π-Aromatic System by a Fused-Bistetrahydrofuran”, J. Med. Chem., 37,
2506-2508 (1994). So far as the position before the priority date was
concerned, the respondents’ evidence drew attention to an article (M. Pezechk et
al, “A new route to perhydro- and tetrahydro- furo-2,3b furans via
radical cyclisation”, Tetr. Letters, 27, 32, 3715-3718 (1986)) which
discloses the heterocyclyloxy portion of the P1 group in darunavir
(but not the whole P1 group) as an intermediate in a reaction scheme
for the synthesis of insect anti-feeding compounds.
The issue
27.
The appellants contend that for the product to be protected by a basic
patent for the purposes of Article 3(a) it must be shown that “the skilled team
would recognise the product as forming a part of the subject matter of the
patent by reference to a careful reading of the patent based on the common
general knowledge at the priority date”. They submit that, given the large
number of compounds covered by the claim and the unusual nature of the P1
substituent on darunavir, that test is not satisfied in the present case. The
respondents disagree and contend that darunavir will be protected by the patent
if it is one of the class of products defined and claimed in the claims of the
patent by reference to the Markush formulae.
The CJEU
jurisprudence on the interpretation of Article 3(a)
28.
The approach to the interpretation of the predecessor to the SPC
Regulation was stated by the CJEU in Case C-482/07 AHP Manufacturing v
Bureau voor de Industriele Eigendom [2009] ECR I-7295 at [27]:
“Next, the Court observes that the second sentence of Article
3(2) of Regulation No 1610/96 must be interpreted not solely on the basis of
its wording, but also in the light of the overall scheme and objectives of the
system of which it is a part (see, by analogy, Case C-292/00 Davidoff
[2003] ECR I-389, paragraph 24).”
29.
The SPC Regulation seeks to strike a balance between the interests at
stake. These interests were identified by Advocate General Trstenjak in her
opinion in Case C-130/11 Neurim Pharmaceuticals (1991) Ltd v
Comptroller-General of Patents [EU:C:2012:268], [2013] RPC 23 as being (1)
the undertakings which pursue costly pharmaceutical research, who favour an
extension of the term of patent protection for the fruits of their research,
(2) the producers of generic medicines who, as a consequence of the extended
term are unable to produce and market generic medicines, (3) patients and the
public who have an interest both in the introduction of new medicines and in
those medicines being available at low prices and (4) State health systems
which share the interest of the patient as well as an interest in preventing
old active ingredients from being brought to the market in slightly modified
form under the protection of certificates but without genuine innovation.
30.
To a patent lawyer, outside the context of the SPC system, the notion of
what is protected by a patent is not a complex or difficult one. To answer the
question of whether a particular product is protected, he or she will ask the
question whether the product falls within the claims of the patent (applying
Article 69 EPC and the Protocol). If so, then its sale or supply will be
preventable by the patentee (as an infringing act applying the appropriate
national rules for infringement). The product is therefore protected against
sales by third parties. In conducting that exercise account is not normally
taken of the presence of other active ingredients which are present in the
accused product in addition to the patented one. Claims are normally
interpreted as specifying what must be present, and as having nothing to say
about what must not be present (although of course this can be made an express
requirement of the claim). Further questions may arise if the doctrine of equivalents
is invoked, but that problem does not arise on the present facts. It is
tolerably clear that this straightforward approach is not correct in the
context of the SPC Regulation. To understand the approach which must be taken,
and the way it has been developed by the CJEU, it is necessary to track through
a number of decisions of that court.
Farmitalia
31.
In Case C-392/97 Farmitalia Carlo Erba Srl [2000] RPC 580,
Farmitalia had obtained a German patent for idarubicin. The claims of the
patent specifically covered idarubicin hydrochloride. Farmitalia had also
obtained a marketing authorisation for idarubicin hydrochloride and applied for
a SPC for “idarubicin and salts thereof including idarubicin hydrochloride”.
The German Patent Office granted a SPC for idarubicin hydrochloride, but
refused to grant one for “idarubicin and salts thereof including idarubicin
hydrochloride”. The Bundesgerichtshof (German Federal Court of Justice)
referred questions concerning the interpretation of Article 3 of Council Regulation
1768/92/EEC (the predecessor to the SPC Regulation) to the Court of Justice.
One question (its second) was as follows:
“According to which criteria is it to be determined whether
the product is protected by a basic patent within the meaning of Article 3(a),
where the grant of a protection certificate is sought for the free base of an
active ingredient including any of its salts, but the basic patent in its
patent claims mentions only the free base of this substance and, moreover,
mentions only a single salt of this free base? Is the wording of the claim for
the basic patent or the latter's scope of protection the determining
criterion?”
32.
The issue thus raised therefore concerned whether the inquiry as to what
was protected was limited to what was “mentioned” in the claims, or whether one
could also include that which would be within the scope of protection, such as
alternative salts to the hydrochloride. The court’s answer was as follows:
“23. By its second question, the Bundesgerichtshof is,
in substance, asking what are the criteria, according to Regulation No 1768/92,
and in particular Article 3(a) thereof, for determining whether or not a
product is protected by a basic patent.
…
26. As Community law now stands, the provisions
concerning patents have not yet been made the subject of harmonisation at
Community level or of an approximation of laws.
27. Accordingly, in the absence of Community
harmonisation of patent law, the extent of patent protection can be determined
only in the light of the non-Community rules which govern patents.
…
29. The answer to be given to the second question
must therefore be that, in order to determine, in connection with the
application of Regulation No 1768/92 and, in particular, Article 3(a) thereof,
whether a product is protected by a basic patent, reference must be made to the
rules which govern that patent.”
33.
Had the matter been left there, one could have concluded that, so far as
the UK is concerned, one would apply Article 69 EPC, if necessary the rules
relating to infringing acts in section 60, and that EU law imposed no
additional requirement. That, however, has subsequently proved not to be the
case, although the nature of the additional requirement has proved somewhat
elusive.
Medeva
34.
In Case C-322/10 Medeva BV v Comptroller-General of Patents, Designs
and Trade Marks [2012] RPC 25, Medeva was the proprietor of a patent for
the preparation of a combination of two antigens known as pertactin and FHA
used in a vaccine against whooping cough. It was claimed that this combination
produced a synergistic effect in vaccine potency. The claims covered the
combination of pertactin and FHA. Medeva obtained marketing authorisations in
respect of vaccines each of which was for immunisation against a number of
diseases in addition to whooping cough, namely, diphtheria, tetanus, meningitis
and polio. The vaccines contained between 8 and 11 different antigens,
including, in each case, pertactin and FHA. Medeva filed five applications for
SPCs in respect of the medicinal products the subject of the authorisations,
all of which were refused by the UK Patent Office. The Comptroller refused four
of the applications on the ground that they did not comply with Article 3(a)
since the patent did not protect the combinations of antigens which were the
subject of the authorisations and were specified in the applications. That
decision was in line with a previous first instance decision of Jacob J in Takeda
Chemical Industries Ltd’s SPC Applications (No 3) [2003] EWHC 649 (Pat); [2004] RPC 3.
35.
Medeva appealed to the Patents Court, but its appeal was dismissed by
Kitchin J (as he then was), ([2010] EWHC 68 (Pat); [2010] RPC 20). Medeva
appealed to the Court of Appeal, which referred the following five questions
concerning Article 3(a) to the CJEU ([2010] EWCA Civ 700, [2010] RPC 27):
“1. Regulation 469/2009 (‘the Regulation’) recognises
amongst the other purposes identified in the recitals, the need for the grant
of an SPC by each of the Member States of the Community to holders of national
or European patents to be under the same conditions, as indicated in recitals 7
and 8. In the absence of Community harmonisation of patent law, what is
meant in Article 3(a) of the Regulation by ‘the product is protected by a basic
patent in force’ and what are the criteria for deciding this?
2. In a case like the present one involving a medicinal
product comprising more than one active ingredient, are there further or
different criteria for determining whether or not ‘the product is protected by
a basic patent’ according to Article 3(a) of the Regulation and, if so, what
are those further or different criteria?
3. In a case like the present one involving a multi-disease
vaccine, are there further or different criteria for determining whether or not
‘the product is protected by a basic patent’ according to Article 3(a) of the
Regulation and, if so, what are those further or different criteria?
4. For the purposes of Article 3(a), is a multi-disease
vaccine comprising multiple antigens ‘protected by a basic patent’ if one
antigen of the vaccine is ‘protected by the basic patent in force’?
5. For the purposes of Article 3(a), is a multi-disease
vaccine comprising multiple antigens ‘protected by a basic patent’ if all
antigens directed against one disease are ‘protected by the basic patent in
force’?” (emphasis added).
36.
Questions 1 and 2 are of particular importance in the present case.
Question 1 was a version of the question asked in Farmitalia. Question
2, whether combinations are special, was new and was the first (but by no means
the last) occasion on which this question had been put.
37.
In her opinion in Medeva Advocate General Trstenjak distinguished
between what she called the “subject matter – or extent of protection” of the
basic patent and its “protective effect”. At [67] she concluded that it was
clear from the literal language of the Regulation (including Article 1(c)
“product as such”) that:
“… a patent for ‘an’ active ingredient or ‘a’ combination of
active ingredients which forms only part of the combination of active
ingredients of a medicinal product cannot constitute a ‘basic’ patent within
the meaning of Article 1(c) of [the SPC Regulation]. That is because on a
literal interpretation, only the combination of active ingredients of that
medicinal product in its entirety, and not the patented part of that
combination, can be described as a product within the meaning of Article 1(b).”
38.
She continued at [68] and [69]:
“68. Nor is that conclusion altered in any way by the
discussion conducted in the main proceedings in the context of Article 3(a) of
Regulation No 469/2009, on the distinction between the subject‑matter – or extent of protection
– and the protective effect of the basic patent. That debate concerns, in
particular, the question whether the fact that an active ingredient which is
the subject‑matter
of a patent is an integral part of a combination of active ingredients and, as
a consequence, that entire combination of active ingredients may not be
produced or placed on the market without the consent of the patent proprietor
(that is the protective effect of the patent) implies that the combination of
active ingredients is deemed to be protected by a patent in force.
69. The decisive consideration in that context is the
fact that the definition of the basic patent in Article 1(c) of Regulation No
469/2009 takes as its basis the subject‑matter of the patent, and not its protective effect.
A basic patent within the meaning of Regulation No 469/2009 must therefore be
understood as one whose subject‑matter
comprises either a product as such, a process to obtain a product or an
application of a product within the meaning of Article 1(b) of Regulation No
469/2009.”
39.
This reasoning is important: the definition of “basic patent” in Article
1(c) is a patent which protects the product “as such”, and a product is the
active ingredient or combination of ingredients of a medicine. It is
comprehensible as a matter of language, therefore, that a patent for an active
ingredient per se could not provide the basis for a SPC for a
combination product, and a patent for a combination of two active ingredients
could not provide the basis for a SPC for one active alone. In both cases
there would be a mis-match and the patent would not protect the product “as such”.
40.
The Advocate General also conducted a teleological interpretation having
regard to the various interests which I have already summarised at [29] above.
She expressed a concern about the potential for abuse if combination SPCs could
be obtained:
“96. If both the combination of active ingredients of a
medicinal product and a patented active ingredient or combination of active
ingredients contained in it could in future be classified as a product within
the meaning of Article 1(b) of Regulation No 469/2009, there would be a risk
that a manufacturer of medicinal products could develop a number of medicinal
products with different combinations of active ingredients on the basis of one
patented active ingredient or combination of active ingredients and place those
products on the market with a time lag in some cases, for the purpose of
optimising the protection under the certificate.
97. An optimised duration – from the point of view of
the manufacturer of medicinal products – of protection under the patent and the
certificate could, for example, be achieved by ensuring that a first medicinal
product with a patented active ingredient is placed on the market as quickly as
possible in order to exploit the already existing patent protection
commercially. Where the procedure for obtaining an authorisation to place the
product on the market has taken longer than five years, the manufacturer of
medicinal products could at the same time apply for a supplementary protection
certificate and declare the complete combination of active ingredients as the
product. He could then attempt to substantiate the protection under patent law
for that product, required under Article 3(a) of Regulation No 469/2009, by
reference to the protective effect of the basic patent for the patented active
ingredient included in the combination of active ingredients. Subsequently, the
manufacturer of medicinal products could place such products with slightly
differing combinations of active ingredients, also including the patented
active ingredient, on the market and, according to the same logic, apply for
new supplementary protection certificates for them, which could then have a
duration of up to five years.
98. In order to prevent such an undermining of the
system of limitation of the duration of the protection conferred by a
certificate provided for in Regulation No 469/2009, Article 3(a) must be
interpreted as meaning that the product within the meaning of that provision is
the same as the product which forms the subject‑matter of the basic patent within
the meaning of Article 1(c).
41.
The Advocate General’s answers to the questions referred were therefore:
"112. In order to answer the first question, as to how
and on the basis of what criteria Article 3(a) of Regulation No 469/2009 is to
be interpreted and applied, it is necessary to start from the principle that a
product within the meaning of Article 3(a) is to be understood as a product
which forms the subject-matter of a basic patent within the meaning of Article
1(c) of the regulation. Whether a product forms the subject-matter of a basic
patent within the meaning of Article 1(c) and whether that product is protected
by a basic patent in force in accordance with the requirement of Article 3(a)
are determined, in principle, according to the rules governing the basic
patent. However, the definition of a basic patent laid down in Article 1(c) of
the regulation precludes combinations of active ingredients which are not the
subject-matter of a basic patent, but nevertheless enjoy patent protection due
to the presence of a patented active ingredient, from being characterised as a
product within the meaning of Article 3(a).
113. Against that background, the first question must be
answered as follows: the condition for the classification of an active
ingredient or combination of active ingredients of a medicinal product as a
product within the meaning of Article 3(a) of Regulation No 469/2009 is that
that active ingredient or combination of active ingredients forms the
subject-matter of a basic patent within the meaning of Article 1(c) of that
regulation. Whether an active ingredient or combination of active ingredients
of a medicinal product forms the subject-matter of a basic patent within the
meaning of Article 1(c) and whether that active ingredient or combination of
active ingredients is protected by a basic patent in force in accordance with
the requirement of Article 3(a) are determined, in principle, according to the
rules governing the basic patent. However, the definition of the basic patent
laid down in Article 1(c) of the regulation precludes use of the protective
effect of the basic patent from being invoked as a criterion for the purpose of
answering the question whether an active ingredient or combination of active
ingredients of a medicinal product forms the subject-matter of a basic
patent."
42.
The Court of Justice also considered questions 1 to 5 together, choosing
to re-phrase them as follows:
“19. By its first five questions, which it is
appropriate to examine together, the Court of Appeal asks, in essence, whether
Article 3(a) of Regulation No 469/2009 must be interpreted as precluding the
competent industrial property office of a Member State from granting a SPC
where the active ingredients specified in the application include active
ingredients not mentioned in the wording of the claims of the basic patent
relied on in support of such an application.
20. While the Latvian, Lithuanian and Portuguese
Governments submit that only the wording of the claims is relevant for the
purpose of determining whether a product is protected by a basic patent in
force within the meaning of Article 3(a) of Regulation No 469/2009, Medeva and
the United Kingdom Government maintain that the concept of a ‘product …
protected by a basic patent in force’ within the meaning of that provision
corresponds to any combination of substances of a medicinal product directly
infringing the patent.”
43.
The court then repeated what it had said in Farmitalia at
[26]-[27] and continued:
“24. It should be noted that Regulation No 469/2009
establishes a uniform solution at European Union level by creating a SPC which
may be obtained by the holder of a national or European patent under the same
conditions in each Member State. It thus aims to prevent the heterogeneous
development of national laws leading to further disparities which would be
likely to create obstacles to the free movement of medicinal products within
the European Union and thus directly affect the establishment and functioning
of the internal market (see Case C 350/92 Spain v Council [1995] ECR I 1985, paragraphs 34 and 35; Case C 127/00 Hässle [2003] ECR I 14781,
paragraph 37; and Case C 482/07 AHP Manufacturing [2009] ECR I 7295,
paragraph 35).
25. Moreover, it should be recalled that Article 5 of
Regulation No 469/2009 provides that any SPC confers the same rights as
conferred by the basic patent and is subject to the same limitations and the
same obligations. It follows that Article 3(a) of the regulation precludes the
grant of a SPC relating to active ingredients which are not specified in the
wording of the claims of the basic patent.
26. Similarly, if a patent claims that a product is
composed of two active ingredients but does not make any claim in relation to
one of those active ingredients individually, a SPC cannot be granted on the
basis of such a patent for the one active ingredient considered in isolation.
27. That approach is also borne out by the second
subparagraph of paragraph 20 of the explanatory memorandum to the proposal for
Council Regulation (EEC) of 11 April 1990 concerning the creation of a
supplementary protection certificate for medicinal products (COM(90) 101 final)
(‘the explanatory memorandum’), which, in so far as concerns what is ‘protected
by the basic patent’, refers expressly and solely to the wording of the claims
of the basic patent. That interpretation also accords with that given in
recital 14 in the preamble to Regulation (EC) No 1610/96 of the European
Parliament and of the Council of 23 July 1996 concerning the creation of a
supplementary protection certificate for plant protection products (OJ 1996 L
198, p. 30), which refers to the need for ‘products’ to be ‘the subject of
patents specifically covering them’.
28. The answer to the first five questions is,
therefore, that Article 3(a) of Regulation No 469/2009 must be interpreted as
precluding the competent industrial property office of a Member State from
granting a SPC relating to active ingredients which are not specified in the
wording of the claims of the basic patent relied on in support of the SPC
application.”
44.
The conclusion in [25] (and repeated in [28]) that Article 3(a) of the
SPC Regulation precludes the grant of a SPC relating to active ingredients
which are not specified in the wording of the claims of the basic patent is
said to follow from Article 5, which provides that any SPC confers the same
rights as conferred by the basic patent and possibly also from the need to
avoid the heterogeneous development of national laws. The first of these
considerations would seem to me to point in the direction of an infringement
test, whilst the second would be satisfied by any test which the CJEU could lay
down for application by all member states. Nevertheless the court’s conclusion
is consistent with the reasoning of the Advocate General.
45.
When the case returned to the Court of Appeal, [2012] EWCA Civ 523;
[2012] RPC 26, the obscurity of the court’s reasoning was the subject of
argument and comment. In his judgment (with which Etherton and Elias LJJ
agreed) Sir Andrew Morritt C recorded a submission of counsel at [28] that if
the phrase "specified in the wording of the claims" in the Court of
Justice’s decision was interpreted more narrowly so as to require the active
ingredients to be expressly named then it would not be possible to grant SPCs
in relation to “Markush” claims and other classes of product such as salts and
antibodies as well as combination products generally.
46.
He went on to say, at [32], that it was nevertheless quite clear that
the Court of Justice had rejected the infringement test. This was clear from
the Advocate General’s opinion, and although the judgment of the court was “not
so clear” the language used was inconsistent with any suggestion that the
protective effect had any relevance to the issue before the court: see [32].
He continued at [33]:
“33.Thus the issue for the national court is to determine
which active ingredients are specified in the wording of the claims. The
ambit of "specified" may range from express naming, through
description, necessary implication to reasonable interpretation. Where on
that scale the dividing line is to be drawn will necessitate further references
in due course in the light of the facts of the cases in which the issue arises.
The problem for Medeva in this case is that wherever the dividing line is to be
drawn the active ingredients relating to vaccines against diphtheria, tetanus,
meningitis and polio are excluded.” (emphasis supplied).
Eli Lilly
47.
In Case C-493/12 Eli Lilly & Co Ltd v Human Genome Sciences Inc
[2014] RPC 21, Human Genome Sciences (“HGS”) was the proprietor of a patent
which disclosed the existence of a novel member of the TNF ligand superfamily
of cytokines called Neutrokine-α. The patent disclosed the structure of
Neutrokine-α, the sequence of its encoding DNA, its tissue distribution,
its expression and the fact that it was a member of the superfamily. HGS had
not found Neutrokine-α by traditional wet-lab techniques, but by
“bio-informatics” or computational biology, i.e. computer-assisted sequence
homology studies. Consequently, the description in the patent specification was
not supported in any way by any data obtained from in vitro or in
vivo studies, but was essentially a prediction based upon what was known
about other members of the TNF superfamily. Claim 13 of the patent effectively
covered any antibody that bound specifically to the full length
Neutrokine-α polypeptide, or its extracellular domain of which there were
potentially very large but in any event unknown numbers. It did not contain
any structural definition or description of an antibody which might function as
claimed.
48.
Eli Lilly had developed its own antibody for use in the treatment of
autoimmune diseases, which it labelled LY2127399. LY2127399 bound specifically
to Neutrokine-α, and in consequence would be an infringement of claim 13
of the HGS patent if the patent was valid and in force. Eli Lilly sought a
declaration that any SPC that HGS might obtain based on Eli Lilly’s marketing
authorisation for LY2123799 would be invalid. Its case was that the claims of
the HGS patent were too broadly framed for it to be possible to regard
LY2123799 as ‘specified’ in the wording of the claims as required by the Court
of Justice’s decision in Medeva. In order for a SPC to be granted on the
basis of HGS’ patent, its claims would have to be significantly more specific.
49.
Warren J referred the following questions to the CJEU:
“1. What are the criteria for deciding whether ‘the
product is protected by a basic patent in force’ in Article 3(a) of [the SPC
Regulation]?
2. Are the criteria different where the product is not a
combination product, and if so, what are the criteria?
3. In the case of a claim to an antibody or class of
antibodies, is it sufficient that the antibody or antibodies are defined in
terms of their binding characteristics to a target protein, or is it necessary
to provide a structural definition for the antibody or antibodies, and if so,
how much?”
50.
Question 3 is new, the other questions having been asked and not
answered either at all or in affirmative terms on a number of previous
occasions. Question 3 raises the question whether a functional definition can
ever “specify” the product. The CJEU gave its judgment without an Advocate
General’s opinion. It dealt with all three questions together, which it
reformulated as follows at [24]:
“By its three questions, which it is appropriate to consider
together, the referring court asks, in essence, whether Article 3(a) of
Regulation No 469/2009 must be interpreted as meaning that, in order for an
active ingredient to be regarded as ‘protected by a basic patent in force’ within
the meaning of that provision, the active ingredient must be identified in the
claims of the patent by a structural formula, or whether the active ingredient
may also be considered to be protected where it is covered by a functional
formula in the patent claims.”
51.
The court recorded at [29] a submission by the Commission that to insist
upon a literal reference to the active ingredient in the claims of a basic
patent would be unduly restrictive. However the Commission’s view was that,
“…for a competent person and on the basis of the general
knowledge of a person skilled in the art, it should be immediately evident from
the claims of a basic patent that the active ingredient for which an SPC is
sought is actually claimed by that patent.”
52.
The court repeated at [31] its previous observation in Medeva (at
[23]) that since no harmonised European Union patent rules are applicable in
the main proceedings, the extent of the protection conferred by a basic patent
can be determined only in the light of the non‑European Union rules governing
patents. It continued at [32]:
“32. It must be borne in mind that the rules for
determining what is protected by a basic patent for the purpose of Article 3(a)
of Regulation No 469/2009 are those relating to the extent of the invention
covered by such a patent, such as the rules laid down in the main proceedings
in section 125 of the UK Patents Act 1977. Where the patent in question has
been granted by the EPO, those rules are also the rules laid down in the EPC
and Protocol on the Interpretation of Article 69 of that convention.
33. On the other hand, as is apparent from the
response given by the Court to questions 1 to 5 in the case which gave rise to
the judgment in Medeva, for the purpose of determining whether a product
is ‘protected by a basic patent in force’ within the meaning of Article 3(a) of
Regulation No 469/2009, recourse may not be had to the rules governing
infringement proceedings, such as, in the main proceedings, those laid down in
section 60 of the UK Patents Act 1977.
“34. By finding that Article 3(a) of Regulation No
469/2009 precludes the grant of an SPC relating to active ingredients which are
not specified in the claims of a basic patent (see Medeva, paragraph 25,
and the orders in Case C-630/10 University of Queensland and CSL
[2011] ECR I 12231, paragraph 31, and Case C-6/11 Daiichi Sankyo [2011]
ECR I 12255, paragraph 30), the Court emphasised the key role played by the
claims for the purpose of determining whether a product is protected by a basic
patent within the meaning of that provision.
…
37. With regard to the fact that the marketing of
that active ingredient by Eli Lilly during the lifetime of HGS’s patent would
constitute an infringement of the patent, it is clear, in the light of what has
been stated at paragraphs 32 and 33 above, that that is not a crucial factor,
for the purpose of granting an SPC on the basis of Regulation No 469/2009,
in particular Article 3(a) of that regulation, in the determination of whether
that active ingredient is protected by that patent.”
53.
The court went on to consider the significance of the fact that
LY2127399 was not mentioned in the patent, and said:
“38. It should be recalled that, in accordance with the
case-law cited at paragraph 34 above, an active ingredient which is not
identified in the claims of a basic patent by means of a structural, or indeed
a functional definition cannot, in any event, be considered to be protected
within the meaning of Article 3(a) of Regulation No 469/2009.
39. With regard to the question whether the use of a
functional definition may alone be sufficient, it should be noted that Article
3(a) of Regulation No 469/2009 does not, in principle, preclude an active
ingredient which is given a functional definition in the claims of a patent
issued by the EPO being regarded as protected by the patent, on condition that
it is possible to reach the conclusion on the basis of those claims,
interpreted inter alia in the light of the description of the invention, as
required by Article 69 of the EPC and Protocol on the interpretation of that
provision, that the claims relate, implicitly but necessarily and specifically,
to the active ingredient in question.
40. With regard to the requirements laid down by the
EPC, it should, however, be noted that the Court does not have jurisdiction to
interpret the provisions of that convention, since, unlike the Member States,
the European Union has not acceded to the convention. The Court cannot,
therefore, provide further guidance to the referring court concerning the
manner in which it is to determine the extent of the claims of a patent issued
by the EPO.
41. Moreover, it should be recalled that the SPC is
designed simply to re‑establish
a sufficient period of effective protection of the basic patent by permitting
the holder to enjoy an additional period of exclusivity on the expiry of that
patent, which is intended to compensate, at least in part, for the delay to the
commercial exploitation of his invention by reason of the time which has
elapsed between the date on which the application for the patent was filed and
the date on which the first MA in the European Union was granted (Case C-229/09
Hogan Lovells International [2010] ECR I-11335, paragraph 50; Case
C-443/12 Actavis Group PTC and Actavis UK [2013] ECR, paragraph 31; and
Case C‑484/12 Georgetown
University [2013] ECR, paragraph 36).
42 As stated in recital 4 in the preamble to Regulation
No 469/2009, the purpose of that additional period of exclusivity is to
encourage research and, to that end, it is designed to ensure that the
investments put into such research are covered.
43. In the light of the objective of Regulation No
469/2009, the refusal of an SPC application for an active ingredient which is
not specifically referred to by a patent issued by the EPO relied on in support
of such an application may be justified – in circumstances such as those in the
main proceedings and as observed by Eli Lilly – where the holder of the patent
in question has failed to take any steps to carry out more in-depth research
and identify his invention specifically, making it possible to ascertain
clearly the active ingredient which may be commercially exploited in a
medicinal product corresponding to the needs of certain patients. In such a
situation, if an SPC were granted to the patent holder, even though – since he
was not the holder of the MA granted for the medicinal product developed from
the specifications of the source patent – that patent holder had not made any
investment in research relating to that aspect of his original invention, that
would undermine the objective of Regulation No 469/2009, as referred to in
recital 4 in the preamble thereto.
44. In the light of the foregoing considerations, the
answer to the questions referred is that Article 3(a) of Regulation No 469/2009
must be interpreted as meaning that, in order for an active ingredient to be
regarded as ‘protected by a basic patent in force’ within the meaning of that
provision, it is not necessary for the active ingredient to be identified in
the claims of the patent by a structural formula. Where the active ingredient
is covered by a functional formula in the claims of a patent issued by the EPO,
Article 3(a) of that regulation does not, in principle, preclude the grant of
an SPC for that active ingredient, on condition that it is possible to reach
the conclusion on the basis of those claims, interpreted inter alia in the
light of the description of the invention, as required by Article 69 of the EPC
and the Protocol on the interpretation of that provision, that the claims
relate, implicitly but necessarily and specifically, to the active ingredient
in question, which is a matter to be determined by the referring court.”
54.
Although it is the Court of Justice’s summary which is the binding
source of law, for convenience I would shortly summarise the conclusions in Eli
Lilly as follows:
i)
The rules for determining whether the product is protected are those
relating to the extent of the invention (in the case of a European patent,
those defined by Article 69 and the Protocol): [32].
ii)
Recourse may not be had to the rules relating to infringement, such as
those in section 60 of the Patents Act 1977: [33].
iii)
The fact that the product infringes is not, therefore, “a crucial”
factor: [37].
iv)
The claims have a key role for the purpose of determining whether a
product is protected by a basic patent within the meaning of Article 3(a):
[34].
v)
An active ingredient which is not identified in the claims by any means
(i.e. either a structural or functional definition) is not protected: [38].
vi)
It is not necessary for the active ingredient to be identified in the
claims of the patent by a structural formula: a “functional formula” will do as
well: [39], but:
vii)
It must be possible to reach the conclusion on the basis of the claims,
interpreted inter alia in the light of the description of the invention,
that the claims relate, implicitly but necessarily and specifically, to the
active ingredient in question: [39], [44].
viii)
It is for the national court to determine the application of this test:
[40], [44].
55.
When the Eli Lilly case returned before Warren J, [2014] EWHC 2404 (Pat); [2015] RPC 8, he did not find the application of the court’s
reasoning to the facts of the case to be straightforward. In the end he
considered that the claim “related to” Lilly’s antibody tabalumab. Lilly’s
application for a declaration therefore failed.
56.
In his judgment in the present case Arnold J expressed disagreement with
the route by which Warren J had come to this conclusion, but not necessarily
with the result.
Two other cases
57.
It is necessary to mention two other cases, although these are not of
such central relevance as Medeva and Eli Lilly. These are Case
C-443/12 Actavis Group PTC EHF v Sanofi [2014] RPC 20, and Case C-577/13
Actavis Group PTC EHF and another v Boehringer Ingelheim Pharma GmbH &
Co KG EU:C:2015:165.
58.
The court’s judgment in Actavis v Sanofi was released on the same
day as its judgment in Eli Lilly. Sanofi was the proprietor of a patent
for the drug irbesartan, an anti-hypertensive. It had been granted a SPC for
irbesartan, optionally in the form of one of its salts. It had also been
granted a SPC for a combination of irbesartan (again optionally in the form of
a salt) in combination with another active, hydrochlorothiazide which was a
diuretic. Claim 7 of the patent was to “irbesartan or one of its salts with
acids or bases”, and claim 20 was to a pharmaceutical composition containing a
composition in accordance with a preceding claim, in association with a
diuretic.
59.
Actavis argued that the combination SPC was invalid on two grounds.
Firstly they contended that the combination of irbesartan and
hydrochlorothiazide was not protected by a basic patent within Article 3(a),
since it was not specified or identified in the wording of the claims.
Hydrochlorothiazide was not mentioned at all in the patent. Secondly they
argued that the combination SPC was invalid because the product had already
been the subject of the SPC for irbesartan. This offended against Article 3(c),
or alternatively, it had been the subject of an earlier marketing authorisation
namely the authorisation of the irbesartan itself, and therefore offended
against Article 3(d).
60.
Arnold J referred the following two questions to the CJEU for a
preliminary ruling:
“(1) What are the criteria for deciding whether “the
product is protected by a basic patent in force” in Article 3(a) of …
Regulation No 469/2009?
(2) In a situation in which multiple products are
protected by a basic patent in force, does Regulation [No 469/2009], and in
particular Article 3(c), preclude the proprietor of the patent being issued a
certificate for each of the products protected?”
61.
The judge also proffered his own test “in the hope that it will assist
the Court of Justice to provide a clear answer this time”. He asked himself
what more it was necessary to show in addition to infringement in order for the
product to be protected. He said at [76]:
“… the answer is that the product must infringe because it
contains an active ingredient, or a combination of active ingredients, which
embodies the inventive advance (or technical contribution) of the basic patent.
Where the product is a combination of active ingredients, the combination, as
distinct from one of them, must embody the inventive advance of the basic
patent.”
62.
The court answered question 2 first, and therefore did not address
question 1. It concluded that Article 3(c) operated to prevent the grant of a
second SPC to Sanofi for the combination. The case is only of significance
because of the court’s use, in connection with a statement of the objective of
the SPC Regulation, of the concept of the “core inventive advance” of the
patent. Thus at [41] the court says:
“It should be recalled that the basic objective of Regulation
No 469/2009 is to compensate for the delay to the marketing of what constitutes
the core inventive advance that is the subject of the basic patent, namely, in
the main proceedings, irbesartan. In the light of the need, referred to in
recital 10 in the preamble to that regulation, to take into account all the interests
at stake, including those of public health, if it were accepted that all
subsequent marketing of that active ingredient in conjunction with an unlimited
number of other active ingredients, not protected as such by the basic patent but
simply referred to in the wording of the claims of the patent in general terms,
such as, in the case of the patent in the main proceedings, ‘beta-blocking
compound’, ‘calcium antagonist’, ‘diuretic’, ‘non-steroidal anti-inflammatory’
or ‘tranquilizer’, conferred entitlement to multiple SPCs, that would be
contrary to the requirement to balance the interests of the pharmaceutical
industry and those of public health as regards the encouragement of research
within the European Union by the use of SPCs” (emphasis added).
63.
In Actavis v Boehringer, Boehringer had a patent which claimed
numerous molecules, including telmisartan and one of its salts. Telmisartan
was an antihypertensive agent marketed by Boehringer and for which they
obtained a SPC for “telmisartan optionally in the form of one of its salts”.
Boehringer also later obtained a SPC for the combination of telmisartan and
hydrochlorothiazide. In order to obtain this second SPC it had amended the
basic patent to include a claim for telmisartan and hydrochlorothiazide. Birss
J referred a number of specific and more general questions to the CJEU, not all
of which the court felt it appropriate to answer. In short the court held that
Articles 3(a) and 3(c) precluded the grant of a second SPC to Boehringer for
the combination:
“36. In the light of the need, referred to, inter alia,
in recital 10 in the preamble to Regulation No 469/2009, to take into account
all the interests at stake, including those of public health, if it were
accepted that all subsequent marketing of an active ingredient in conjunction
with an unlimited number of other active ingredients which do not constitute
the subject-matter of the invention covered by the basic patent would confer
entitlement to multiple SPCs, that would be contrary to the requirement to
balance the interests of the pharmaceutical industry and those of public health
as regards the encouragement of research within the European Union by the use
of SPCs (see, to that effect, judgment in Actavis Group PTC and Actavis UK,
EU:C:2013:833, paragraph 41).
37 Accordingly, in view of the interests referred to in
recitals 4, 5, 9 and 10 in the preamble to Directive 469/2009, it cannot be
accepted that the holder of a basic patent in force may obtain a new SPC,
potentially for a longer period of protection, each time he places on the
market in a Member State a medicinal product containing, on the one hand, an
active ingredient, protected as such by the holder’s basic patent and
constituting the subject-matter of the invention covered by that patent, and,
on the other, another substance which does not constitute the subject-matter of
the invention covered by the basic patent (see, to that effect, judgment in
Actavis Group PTC and Actavis UK, EU:C:2013:833, paragraph 30).
38 It follows that, in order for a basic patent to
protect ‘as such’ an active ingredient within the meaning of Articles 1(c) and
3(a) of Regulation No 469/2009, that active ingredient must constitute the
subject-matter of the invention covered by that patent.
39 In the light of the foregoing considerations, the
answer to Questions 2 and 3 is that Article 3(a) and (c) of Regulation No
469/2009 must be interpreted as meaning that, where a basic patent includes a
claim to a product comprising an active ingredient which constitutes the sole
subject-matter of the invention, for which the holder of that patent has
already obtained an SPC, as well as a subsequent claim to a product comprising
a combination of that active ingredient and another substance, that provision
precludes the holder from obtaining a second SPC for that combination.”
64.
The case therefore states the condition for a basic patent to protect
the active ingredient as such as being that the active ingredient must
constitute the subject matter of the invention covered by the patent. That test
is the same as that advanced by the Advocate General in paragraph 113 of her
opinion in Medeva.
Pending
references
Teva v Gilead
65.
In Teva UK v Gilead Sciences Inc [2017] EWHC 13 (Pat), Arnold J
made a further reference to the CJEU. In that case the SPC was for a
combination product consisting of two active ingredients, namely (i) tenofovir
disoproxil (“TD”) in the form of the fumarate (“TDF”) and (ii) emtricitabine
(also known as FTC) in a single, fixed dose tablet. TD and emtricitabine were
both inhibitors of a viral enzyme known as reverse transcriptase. Gilead’s
patent claimed a class of compounds by reference to Markush formulae, which
included TD. There was no reference in the patent to emtricitabine. It did
however contain a subsidiary claim, claim 27, in the following terms:
“A pharmaceutical composition comprising a compound according
to any one of claims 1-25 together with a pharmaceutically acceptable carrier
and optionally other therapeutic ingredients.”
66.
Teva contended that the words “other therapeutic ingredients” did not
specify any active ingredient, whether structurally, functionally or otherwise.
On the contrary, they covered a virtually unlimited range of active ingredients
for the treatment of many diseases. Indeed, emtricitabine was not approved for
clinical use until seven years after the priority date of the patent and there
was no evidence that it was known to be efficacious at that date. Gilead
disagreed and contended that all that was necessary was that emtricitabine fell
within the scope of protection of claim 27 of the patent applying Article 69
EPC and the Protocol.
67.
Both sides contended that the law was now sufficiently clear that a
further reference to the CJEU was not necessary. The judge disagreed, and
referred the following question to the CJEU:
“What are the criteria for deciding whether ‘the product is
protected by a basic patent in force’ in Article 3(a) of the SPC Regulation?”
68.
Although the question is asked in general terms, it is plain from the
arguments of the parties and the judgment that the uncertainty which gives rise
to the need for a reference is the breadth of the definition of the “other
therapeutic ingredients” in the claim, and whether such a broad definition can
be sufficient for the purposes of Article 3(a). The judge also proffered again
his own suggested test, first offered in Actavis v Sanofi (see paragraph
[61] above) in case it might assist the CJEU to “to provide a clear answer this
time”.
The German
sitagliptin reference: Decision 14W (pat) 12/17
69.
Shortly before the hearing we were informed that, in Decision 14 W
(pat) 12/17 dated 17 October 2017, the Bundespatentgericht (German Federal
Patents Court, BPatG) had referred a question to the CJEU in a case which, like
Eli Lilly, concerned a functional claim. The patent claim related to an
“activity lowering effector [of DPP IV] for use in lowering the blood glucose
level” in mammals. The invention was said to be useful in the treatment of
diabetes. I will refer to this case as “Sitagliptin”.
70.
The active substance sitagliptin was developed after the date of the
patent. The German Patent Office rejected the application for the SPC on the
basis that the product was defined in the patent purely functionally, and that
the subject matter of the patent did not extend to the subsequently developed
product, sitagliptin. The appellant appealed on the basis that the
contribution and core value of the patented contribution was not in specific
compounds but in the use of DPP IV inhibitors generally. Sitagliptin was
precisely such a DPP IV inhibitor and fulfilled the functional definition of
the claimed class of active substances. An “individualised disclosure” was not
necessary. It also relied on the decisions in Actavis v Sanofi and Actavis
v Boehringer, as well as Arnold J’s “inventive advance” test as proposed in
the present and earlier cases to support a submission that the essential
question was whether the product in question is an embodiment of the inventive
concept of the patent.
71.
The BPatG’s own view (see paragraph [3.3]) was that Actavis v Sanofi
and Actavis v Boehringer did not affect the test set out in Eli Lilly.
The court stated, at [4] and [5], its understanding of the case law of the CJEU
in Medeva and Eli Lilly. At [5] it said that it considered
decisive
“that the product in question is described in the claims of
the basic patent in such a specific way that it forms part of the protected
subject-matter of the patent claims. Article 69 is not only concerned with
determining the extent of protection, but distinguishes between the
determination of the subject-matter of the claims required in an initial
examination step, on the one hand, and the determination of the extent of
protection for this subject-matter as relevant to the question of infringement
on the other hand”.
72.
The court considered (see paragraph [6]) that the requirements laid down
by the Court of Justice were only met if the active ingredient in question “is
specified in the claims in such a way that it can be identified as such and it
is actually provided to the skilled person.” The court, at [8], expressly
rejected the invitation to adopt a “core inventive contribution” test proffered
in the cited English cases, which it considered had its place only in
connection with Article 3(c). Perhaps more importantly, at [10], the court
indicated that it considered the facts of the case now before us to be
“absolutely comparable to those in the present case”:
“This is because Darunavir, the substance at issue in the
British decision, (like an almost endless number of other compounds) falls
under a Markush formula contained in the claims of the basic patent, while in
the present case the active substance sitagliptin (like an incalculable number
of other compounds), falls under a functional definition which is contained in
the patent claims.”
73.
Accordingly the BPatGhas decided to refer questions to the CJEU. One of
the reasons it chose to do so was the existence of a differing proposed test in
the United Kingdom, as well as a divergence of practice at the level of the
granting authorities in other member states. The court’s questions are:
“1. Is a product protected by a basic patent in force
according to Article 3(a) of [the SPC Regulation] only if it belongs to the
protected subject-matter as defined by the patent claims and is thus provided
to the person skilled in the art as a specific embodiment?
2. Is it therefore not sufficient for the requirements of
Article 3(a) of [the SPC Regulation] that the product in question meets the
general functional definition of an active substance class as mentioned in the
claims, but beyond this does not constitute a specific embodiment of the method
protected by the basic patent?
3. Is a product consequently not protected under Article 3(a)
of [the SPC Regulation] by a basic patent in force even if it is covered by the
functional definition contained in the claims, but was developed based on
independent inventive activity only after the basic patent application was
filed?”
74.
The first two questions are, in effect, based on the theory that the
active ingredient must be provided to the skilled person as a specific
embodiment. That theory appears, in effect, to demand an actual disclosure of
the active ingredient.
The judgment of
Arnold J
75.
The judge dealt with the law relatively briefly, as he had previously
summarised it in Teva UK v Gilead (cited above). The judge observed at
[61] that the broadest tenable interpretation of Article 3(a) was that it was
sufficient that the product fell within at least one claim of the patent
applying the rules relating to the extent of protection as ascertained by the
application, in the case of a European patent, of Article 69 EPC and the
Protocol. He recognised, however, that at present it did not appear that that
interpretation was correct, because the CJEU has so far held that more is
required.
76.
The CJEU jurisprudence had initially indicated that it was necessary for
the product to be “specified” or “identified” in the wording of the claims.
Subsequently, however, the CJEU had held that it was not necessary for the
active ingredient to be identified in the claim by means of a structural
formula: and that it was sufficient for the active ingredient to be covered by
a functional description provided that the claims “relate, implicitly but
necessarily and specifically, to the active ingredient”. It was clear from this
that the identification of the active ingredient in the claim by means of a
structural formula is permissible, but not essential; that it was not necessary
for the claim individually to name or depict the active ingredient; and that it
was not necessarily an objection that the claim in question covers a large
number of other compounds in addition to the active ingredient in question.
77.
For those reasons the judge held at [64] that it was sufficient for the
claim to “specify the product by means of a Markush formula which covers it”
(at least without resort to equivalents). On that basis darunavir was
“protected” by the patent.
78.
The judge said that he nevertheless remained of the view which he had
expressed in both Actavis v Sanofi and Teva v Gilead that a
better test would be one which requires that the product fall within the claim
and that it should embody the inventive advance (or technical contribution) of
the claim. If that test was applied to the facts of the present case, the
answer was clear. The inventive advance (or technical contribution) of claim 1
of the patent lay in the identification of the compounds covered by claim 1 as
having utility as HIV protease inhibitors. Darunavir embodies that inventive
advance.
79.
On all tests considered thus far, darunavir was a product protected by
the patent. The judge turned to the appellants’ test at [67]. As to that he
made two points of importance for present purposes. Firstly, the test was
really a breadth of claim test. It was not the function of patent offices when
assessing applications for the grant of SPCs to have to consider whether the
breadth of the claims of the basic patents relied on is justified. That would
not make for a simple and transparent system, as envisaged in paragraph 16 of
the Memorandum. By contrast, paragraph 39 of the Memorandum did envisage SPCs
being granted where “a patent protects a series of products based on the same
formula”. Secondly he said that it was implicit in the appellants’ case that
some compounds covered by claim 1 of the patent were “protected” by the patent,
while others were not; but it was wholly unclear where and how the line between
the two groups of compounds was to be drawn.
80.
The judge considered that, in reality, the Claimants’ objection was that
the claims of the patent were of excessive breadth because they encompassed a
vast number of compounds, of which the skilled person could not make even a
tiny fraction, and which it is not plausible would all be efficacious as
protease inhibitors. If well founded, however, that was an objection to the
validity of the patent. It amounted to saying that the claims were obvious on
AgrEvo grounds or insufficient. However, the appellants had not put the
validity of the patent (as opposed to the SPC) in issue, and this collateral
means of challenging validity was not open to them.
81.
The judge therefore held that there was no tenable construction of
Article 3(a) which led to the conclusion that darunavir was not protected by
the patent.
The arguments on
the appeal
82.
Ms May submitted, firstly, that the judge had made “a fundamental error
of principle” in not referring questions to the Court of Justice. In her
written submissions she said it simply was not open to the judge not to refer
the matter to the CJEU. I do not think this is a productive line of argument,
however. The real question for us is whether the judge’s self-direction on the
applicable law is correct, and whether he then applied the law correctly to the
facts. If he did so the appeal will fail. If he did not do so, we can either
allow the appeal, or if the law is uncertain, refer a question ourselves to the
CJEU. The judge’s decision to refer or not to refer does not give rise to an
independent ground of appeal.
83.
The respondents submitted that a reference would be futile, given that
there is no real possibility of the answer to such a reference being received
and dealt with in this court before the expiry of the SPC. They did not,
however, submit that the appeal was otherwise an abuse of process, on the
grounds, for example, that it was now wholly academic. It follows that we are
obliged to engage with the issues on the appeal. If, we consider that a
reference to the CJEU is necessary to enable us to decide those issues, it must
follow that a reference is not futile.
84.
More significantly, Ms May submitted that the judge had wrongly analysed
the Medeva and Eli Lilly decisions. Medeva had laid down
a general test for when an active ingredient was protected by a basic patent:
it had to be specified in the wording of the claim. That test applied to
combination claims as well as to claims for a single class of active
ingredients. It had, however, given rise to the question of how specific the
claim must be. The Eli Lilly case had attempted to answer that
question, but had not provided a complete answer. The requirement that the
claims relate “implicitly, but necessarily and specifically” to the product
applied to Markush claims but remained unclear. The judge had failed to give
weight in his analysis of the Eli Lilly judgment to the need for the
claim to relate to the active ingredient “necessarily and specifically”. The
first half of paragraph [43] of the Eli Lilly judgment contained an
indication as to what was meant by this phrase, by indicating that it was
relevant if the patentee had not carried out “more in-depth research” to
“identify his invention specifically, making it possible to ascertain clearly
the active ingredient which may be commercially exploited in a medicinal
product corresponding to the needs of certain patients”.
85.
Ms May submitted that, if the judge had grappled with the Eli Lilly test,
he should have concluded that, because of the unusual nature of the P1
substituent group, darunavir was not specified implicitly, but necessarily and
specifically in the claims of the patent. Darunavir was just one of a vast
number of compounds encompassed by the claims of the patent. Further, even if
one could contemplate writing down the entire list, darunavir would not be on
it because the specific P1 group employed in darunavir was not part
of the common general knowledge of the skilled person.
86.
Ms May next submitted that the judge’s inventive advance test was wrong
in principle, at least when applied to a Markush claim. In the case of a
Markush claim, the default position was that every compound which fell within
the formula in terms of its structure would also have the claimed activity, and
therefore embody the inventive advance. The inventive advance test therefore
added nothing to an infringement test in the case of this particular class of
claim. Yet the CJEU had held that “something more” than infringement was
required before the product could be deemed protected by the basic patent.
87.
Ms May also took issue with the way the judge applied the inventive
advance test to the facts of this case. There was evidence in the case, which
the judge treated as irrelevant, that some of the compounds covered by the
claim were difficult to synthesise or unstable. Accordingly this was a case
where the inventive contribution was not commensurate with the breadth of the
Markush claim. Ms May went further and invited us to make a finding that
darunavir did not fall within the inventive contribution of the patent, because
of the unusual nature of the P1 group. A product which the skilled
person cannot identify from the patent cannot be taken to be part of the
inventive advance of that patent.
88.
Finally, Ms May submitted that the judge had failed to adopt the
appellants’ test, which was a test that struck a fair balance between the
extremes of the infringement test on the one hand and a disclosure test on the
other. The appellants’ test gave rise to a protected class of products which
was narrower than the scope of the claim, because it was limited to the
products which the skilled person could envisage based on the common general
knowledge. It was not, however, as narrow as a specific disclosure test, which
Ms May disclaimed before us, as she had done before the judge. The correct
approach was to tell the skilled person the structure of the product which is
to be the subject of the SPC, and to ask whether that product was one which he
or she could have identified at the priority date from a careful reading of the
patent using common general knowledge.
89.
Mr Mitcheson submitted that the appellants’ test was unworkable, and
contrary to the simple, transparent, easy to administer and objective test foreseen
in the Memorandum. Instead of the relatively straightforward test of whether
the Markush formula encompassed the product in question, it required national
patent offices to conduct an evidence-based enquiry into whether the
substituent groups not actually disclosed in the specification were or were not
part of the common general knowledge. That was not a test which had thus far
been adopted in relation to Markush claims. Indeed the SPC for darunavir had
been granted in all 16 designated states of the patent, including Germany.
90.
Mr Mitcheson also submitted that the effect of the test propounded by
the appellants was to discriminate against a phase of medical research, namely
the phase in which the structure-activity relationship has been discovered but
the individual specific compound which was to be administered to humans had not
yet been singled out. This was contrary to the objectives spelled out in the
Memorandum that all types of medical research were to be incentivised.
91.
Mr Mitcheson identified what he called a spectrum of specificity. In Medeva
there was nothing at all in the claim to identify the additional
ingredient, beyond the use of the word “comprising” which allowed for
additional ingredients for the purposes of determining infringement. Then there
were claims which actually called for “other therapeutic ingredients”, such as
that in Teva v Gilead. That was still an entirely generic description,
but it was a suitable candidate for a reference to the CJEU because it was more
specific than Medeva. Then, in Eli Lilly, the claim had a
functional definition of an antibody. It was plain that this definition did
not, indeed could not, define the antibody structure precisely, but the CJEU
did not reject this mode of definition outright. The national court had
subsequently upheld the validity of the SPC. Finally there was a claim of the
kind which was the subject of the Sitagliptin reference. That claim was
also functional, but did not tell you whether the “effector” was an antibody, a
small molecule or a biomolecule. This was less defined than the claim in Eli
Lilly, which made a reference an appropriate course.
92.
The present case did not throw up the problems of specificity which
arose in Eli Lilly and in the German reference (which were both
functional claims, the German claim being even less specific than the claim in Eli
Lilly). The present case defined a class of compounds by both function and
structure. It was not necessary in a case such as the present, where the
active ingredient was defined in the claims by means of a structural formula,
to look for further specificity in the claims. There was no unanswered
question which had to be referred to the CJEU. That was illustrated by the fact
that a number of national offices had rejected the SPC which was the subject of
the Sitagliptin reference, whilst all national offices asked to do so
had granted the daunavir SPC the subject of the present case.
93.
So far as the Sitagliptin reference was concerned, Mr Mitcheson
submitted that the German court had misunderstood the basis on which Arnold J
had decided the present case. The court appeared to have understood that Arnold
J had decided the case on the basis of the core inventive advance test. That
was not the case, however. The ratio of Arnold J’s decision was that there was
no tenable test on which darunavir could be held not to be protected by
the patent. Further, it was not the case at all, as the German court had
suggested, that the facts of the two cases were indistinguishable.
94.
Paragraph [43] of the Eli Lilly decision was concerned with the
situation where a third party had obtained a marketing authorisation for a
specific product not mentioned in the basic patent. That had originally been
an issue in the reference, and was therefore the subject of evidence and
submissions, but Eli Lilly had dropped it before the hearing. Moreover
in the Eli Lilly case the CJEU must have implicitly rejected Lilly’s
case that the criteria for determining whether a product is protected involved
inquiring into the breadth of the claim. That was effectively the same test as
was now propounded by the appellants here.
95.
Mr Mitcheson further submitted that, although it was correct that the
Markush formula in the present case defined a large number of compounds, the
presence of the mandatory structural backbone in fact meant that it defined a
fairly limited chemical space.
96.
Mr Mitcheson submitted, finally, that the problem of abuse identified by
the Advocate General in Medeva concerning the grant of successive SPCs
for a patented active ingredient in combination with a variety of other
ingredients had been dealt with by Article 3(c) in Actavis v Sanofi and
Actavis v Boehringer.
Discussion
97.
I will refer to the CJEU’s requirement, formulated for the first time in
Eli Lilly, that, in order to be protected by the basic patent, the claim
must relate to the product implicitly, but necessarily and specifically as “the
Lilly requirement”. If it were possible to say that the Lilly requirement
is limited to functional claims, or alternatively, if it applies to all
claims, that it is always satisfied by a Markush claim which covers the active
ingredient, then it would follow that the appeal must be dismissed. If the
position is not clear, however, we may have to refer a question to the CJEU.
98.
An important point of detail is the time at which and the circumstances
in which the national authority has to determine whether a product is protected
by a basic patent. The appellants do not suggest that the exercise for the
skilled person of determining whether a product is protected by a basic patent
should be performed in ignorance of the product in question. The judge
accepted the respondents’ submission that the question whether a product is
protected by claims in a basic patent falls to be judged when the product is
known, and when it has been authorised to be placed on the market as a
medicinal product. I did not understand Ms May to challenge that proposition.
I consider it to be correct. That conclusion still leaves open the question of
what is the necessary exercise for determining whether the product is protected
by the patent. The two candidates which remain are (a) asking whether it is
clear that the product is claimed as such; and (b) asking whether the
product is one which is sufficiently identified. A test which goes
further and asks whether the active ingredient is specifically disclosed is not
advanced by the appellants.
99.
One would have thought that the choice between these two remaining
alternatives could be made according to what it is that the Lilly requirement
is seeking to achieve. If the object of the requirement is to ensure that the
product is an embodiment of the inventive effort or advance contributed by the
patentee, then the first of the two candidates would seem to be the appropriate
test. On the other hand, if the object of the requirement is to ensure that
the patent demonstrates that the proprietor has in fact come close to an actual
realisation of the product, then the second of the two candidate tests would
seem to be more appropriate. Indeed one could go further and insist on an
actual disclosure of the active ingredient in question. Even then, given the
ability to generate chemical structures by computer technology without wet-lab
techniques, the mere fact that a structure is precisely specified in a patent
may be a poor indicator of whether the patentee has in fact actually made it or
performed any research on it.
100.
The Lilly requirement stems from the CJEU's decision in Medeva
that all members of a combination of active ingredients which is the subject of
a SPC must be specified in the wording of the claims. The requirement follows,
at least according to the Advocate General's reasoning, from the fact that the
product the subject of the SPC is to be protected "as such" by the
basic patent. Medeva was not a case about how closely each component of
the combination needs to be specified in the claims. Rather it was a case
which ruled out a SPC where some of the products were not specified in the
claims at all. It was a case in which one had a plain mis-match between the
basic patent and the SPC. If there is a mis-match between the product the
subject of the SPC and that protected as such by the patent, one can see how
infringement rules could lead to an incorrect result. That is because the
infringement rules do not require matching in this way. A claim to a single
active ingredient is infringed by a combination of that product and
another active: but the patent does not protect the combination product as
such.
101.
In the case of a SPC with a single active ingredient, the reasoning in Medeva
requires that the basic patent protect that active ingredient as such. The
reasoning is not informative as to how specifically the claims must focus on
the active ingredient, or what underlies the requirement that they should do
so.
102.
It is not clear to me that the CJEU's judgment in Eli Lilly takes
the matter much further. Eli Lilly was specifically concerned with
functional claims. Functional claims and structural claims are
fundamentally different in terms of what they require the skilled person to do
in order to determine whether a particular product is specified by a claim. A
structural claim simply requires one to read the claim and the specification,
look at the structure of the product and decide whether it is a product
specified in the claims. Functional claims, by contrast, require one to
perform a functional test on the product. For example a claim to an antibody
by reference to a binding ability normally requires one to perform a practical
test to determine whether the antibody actually binds to something else.
103.
Medeva therefore left a substantial unanswered question as to
whether a product could be specified by a functional claim at all. It might be
thought that it was simply too difficult for a patent office, operating the
simple, transparent and objective system set up by the SPC Regulation, to work
out whether a particular antibody was one specified in the claim. Problems of
that nature do not arise in the case of Markush claims.
104.
The submission by the Commission recorded at paragraph 29 of the
judgment in Eli Lilly (see paragraph [51] above) that “on the basis of
the general knowledge of a person skilled in the art, it should be immediately
evident from the claims of the basic patent, that the active ingredient for
which a SPC is sought is actually claimed by that patent” can also be seen to
be directed at the problem of linking a functional limitation in a claim to a
given product. The Commission was not necessarily urging the court to impose a
new standard of specificity in claims which already define a class of compounds
by reference to a Markush formula. Indeed the Commission accepted that a
literal reference was not required. The submission is expressed in language (“evident
from the claims of a basic patent that the active ingredient … is actually
claimed”) which is consistent with the first of the candidate tests I have
outlined above.
105.
Such help as the judgment in Eli Lilly gives as to what underlies
the specificity requirement is to be found, not in its core reasoning, but in
paragraph [43] of the judgment. That paragraph appears to be one designed to
give the national court assistance in arriving at its judgment in the main
proceedings. It is true that that paragraph is in the context, additionally,
of an application for a SPC based on a third party’s marketing authorisation.
But the first part of the paragraph seems to me to indicate, albeit without
great clarity, that the court considers that at least one way of preventing or
hindering the marketing authorisations of third parties from being used as the
basis for SPCs is to insist on a high degree of specificity in the basic
patent. That might help to prevent a patentee spreading the net in his patent
claims widely and unspecifically, and subsequently fastening on a competitor’s
successfully marketed drug to obtain an extended term which he has not earned.
That is a consideration which does not only arise in the context of functional
claims, and lends force to the suggestion that the requirement for a high
degree of specificity is a general one.
106.
If it is right that there is a general requirement that the active
ingredient which is the subject of the SPC must be identified, the question
arises of how specific the claims must be. I agree with Mr Mitcheson that
there is a spectrum of specificity indicated by the factual scenarios in the
various decided cases and references. I would regard it as plain that a Markush
claim can in some circumstances amount to a sufficiently precise claim for
the purposes of Article 3(a), for example where individual substituents are
identified in the specification, or where classes of such substituents are set
out, and the skilled person would be able to determine the extent of those
classes. However I do not think one can extract from the reasoning in Eli
Lilly the proposition that an active ingredient is adequately identified by
a Markush formula however broadly that formula is framed and however obscure
the particular substituent required to form the active ingredient the subject
of the SPC. I think it is at least arguable that that substituent must be
amongst those which the skilled person would be able to identify based on his
common general knowledge at the priority date. I say so for two reasons.
107.
My first reason for considering that proposition to be arguable is the
insight which paragraph [43] of Eli Lilly gives into the CJEU’s thinking
concerning the purpose of the requirement for the active ingredient to be
identified. If the objective is to ensure that the patent proprietor has come
close to an actual realisation of the product, then the fact that the relevant
substituents cannot be arrived at from a reading of the specification and the
common general knowledge may be highly relevant.
108.
My second reason for considering that proposition to be arguable is the
view expressed by the BPatG in the Sitagliptin reference that the
functional formula in that case and the Markush formula in the present case are
factually indistinguishable for the purposes of Article 3(a). Whilst I
consider that there are significant points of distinction relevant to Article
3(a) between the two classes of case, the BPatG does not agree. It would
therefore appear likely that a German court would take the view that a Markush
formula may, at least in a case like the present, fail to provide protection
within the meaning of Article 3(a). A decision by this court that a Markush
formula will always be adequate for that purpose would therefore lead to
conflicting decisions at least in these two member states.
109.
Like the judge, however, I am concerned with what I see as a fundamental
defect with the “identification” test. The CJEU jurisprudence to date seems to
take it as read that a claim can identify active ingredients with specificity.
However that is not the function of claims in patents. Instead, claims are
concerned with setting the limits to the monopoly. A further defect of the
focus on the claim is that claims can be manipulated by skilful drafting to
protect combinations, without distinguishing between genuine combinations of
products which work together in a new and advantageous way so as to constitute
an inventive advance, and mere collocations of products giving rise to their
separate individual effects. I agree with the judge that a far better test
would be to ask whether the product the subject of the SPC embodies the core inventive
advance of the basic patent.
110.
I think Ms May is wrong when she submits that the core inventive advance
test is inadequate because it imposes no greater requirement in the case of a
Markush claim than would be imposed by an infringement test. That submission is
based on the false premise that any test one proposes, when applied to a
particular type of claim, must add something to a test of infringement. I do
not follow why that should be so. If the objective behind the Lilly requirement
is understood to be that the active ingredient must embody the inventive
advance, then that objective is satisfied by a valid Markush claim. Every
compound encompassed by the claim delivers the core inventive advance. In
other types of claim the test will not be satisfied. To take an example based
on the facts of Medeva, if the vaccine the subject of the SPC did not
take advantage of the synergistic effect in vaccine potency of the combination,
for example by using normal doses, then it would not embody the core inventive
advance.
111.
The adoption of the core inventive advance test remains a possibility
given the pending references from in Sitagliptin and Teva v Gilead,
and the fact that it is becoming clear (see Actavis v Novartis, Actavis v Boehringer)
that the possible abuse identified by the Advocate General in Medeva can
be dealt with through Article 3(c). If that test were adopted across the board
and applied here, despite Ms May’s submissions concerning its application
(which I reject), I have no doubt that the SPC would satisfy Article 3(a).
112.
By recognising the common general knowledge test as arguable I am not to
be taken as suggesting that there are not some very undesirable consequences if
this approach were to be adopted. Principal amongst these is the difficulty
the test would present to patent offices around Europe in its application. It
is a very long way indeed from the simple, transparent and objective approach
foreseen by the Memorandum. I also agree with Mr Mitcheson that there is a
danger that too narrow an approach to what is protected will discriminate
unfairly against certain stages of pharmaceutical research, contrary to the
objectives in the memorandum, although it might fairly be said that this raises
an issue of policy.
113.
I do not, however, accept that the appellants’ common general knowledge
test is essentially a breadth of claim test, if by that it is meant that it is
a collateral attack on the validity of the patent. It is simply a test of
whether the claim meets the requirement that the active ingredient be
identified specifically.
114.
In case it would assist the Court of Justice I will express my
provisional conclusion. Left to myself, I would have concluded that darunavir
was a product protected by the claims of the patent. In the case of a product
with a single active ingredient and a patent with a claim which identifies a
number of compounds by means of a Markush formula, all of which compounds
embody the core inventive technical advance of the patent, the test should be
whether the skilled person, considering the claims of the patent on the one
hand and the structure of the product in question on the other, would
immediately recognise that the active ingredient in question is one of those
specified by the formula. On the facts of the present case as found by the
judge, that test is satisfied. However, for the reasons I have given, it is
not clear that this is the correct approach in EU law.
115.
I would therefore propose that this court should stay the present appeal
proceedings and refer the following question to the CJEU:
“Where the sole active ingredient the subject of a
supplementary protection certificate issued under [the SPC Regulation] is a
member of a class of compounds which fall within a Markush definition in a claim
of the patent, all of which class members embody the core inventive technical
advance of the patent, is it sufficient for the purposes of Article 3(a) of the
SPC Regulation that the compound would, upon examination of its structure,
immediately be recognised as one which falls within the class (and therefore
would be protected by the patent as a matter of national patent law) or must
the specific substituents necessary to form the active ingredient be amongst
those which the skilled person could derive, based on their common general
knowledge, from a reading of the patent claims?”
116.
If my Lords agree I would invite the parties to seek to agree a draft
order for reference. In the event that they cannot agree we will give
directions for submissions in writing.
Lord Justice
Kitchin
117.
I agree.
Lord Justice
Lewison
118.
I also agree.